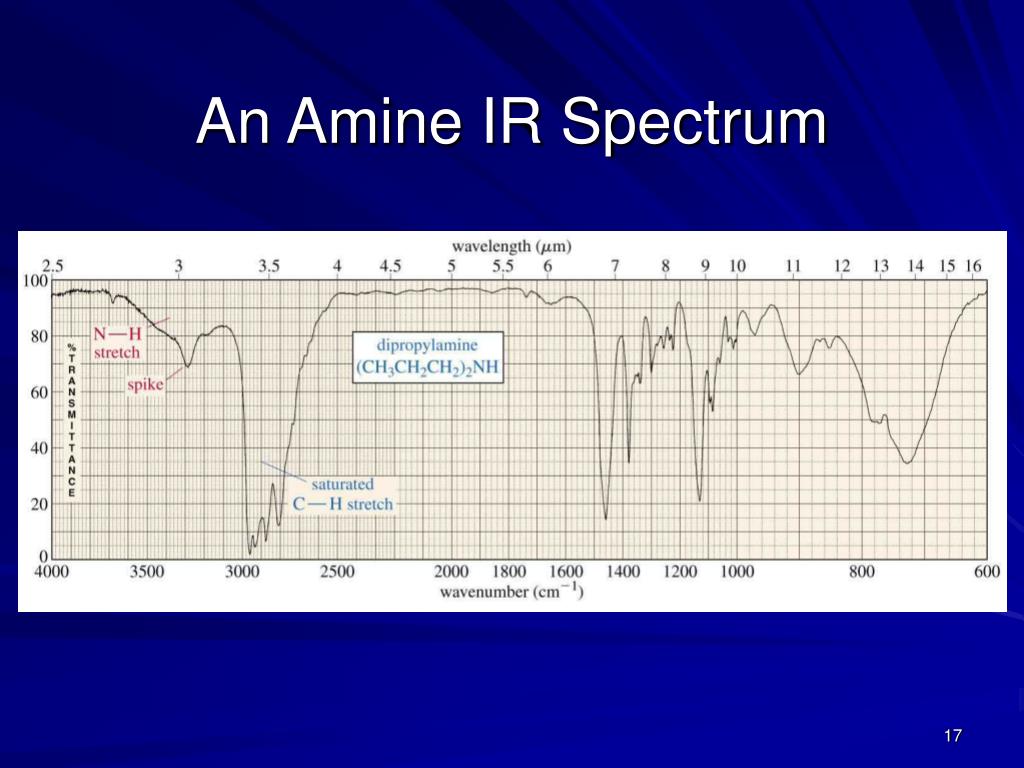 
Label 1-2 relevant peaks that will help you distinguish these two functional groups.How would the spectrum of 2-octanone differ from 1-octanal?.This peak usually has two bands around 28 cm −1. Find the sp 3 C-H stretches and C-C stretches in the spectrum below.Īldehydes also have an sp 2 C-H stretch due to the aldehydic H.Describe the difference.Īldehydes, such as 1-octanal, have sp 3 C-H stretches and C-C stretches.Īll carbonyl functional groups have a distinctive C=O stretch that absorbs strongly around 1700 cm -1. The N-H stretch (or two) above 3200 cm -1 has a significantly different shape than the alcohol O-H stretch.They also have an N-H stretch (or two) above 3200 cm -1.Let’s consider the spectrum of 1-hexylamineĪmines have sp 3 C-H stretches. They lack a _ stretch that is present in an alcohol.Īmines are similar to alcohols, also.Find these key peaks in the spectrum below.Here is the spectrum of butyl ether.Įthers have sp 3 C-H stretches. The C-O stretches show up between 1000-1300 cm -1.Įthers are similar to alcohols. The shape of this OH peak is very distinctive.Most OH stretches show up between 3200-3400 cm -1. There are two new stretches: O-H and C-O. Find the sp 3 C-H and the C-C stretches in the spectrum below. 
Can you tell how many C-C bonds or C=C are in a structure from the IR spectrum?.The spectra of two different alkenes are shown below. Based on the location of the sp 3 C-H in octane, which of these is the new C=C? There are two different types of C-C stretches.Based on the location of the sp 3 C-H in octane, which of these is the new sp 2 C-H? There are two different types of C-H stretches: sp 3 C-H and sp 2 C-H.Based on Hooke’s Law which peak corresponds to C-H stretches? Which peak corresponds to C-C stretches?.How many peaks would you expect to see?.List the different types of bonds present in this structure.* All spectra are either from SDBS (Japan National Institute of Advanced Industrial Science and Technology) or simulated. IR can be a useful tool for determining what types of bonds (functional groups are present in a sample. Infrared Spectroscopy: Functional Group Determination* IR can therefore be a useful tool for determining what types of bonds (functional groups are present in a sample. Hint: consider atomic weights of halides. Many different vibrations, including C-O, C-C and C-N single bond stretches, C-H bending vibrations, and some bands due to benzene rings are found in this region. This region is notable for the large number of infrared bands that are found there. *IR absorbance is typically measured in wavenumbers (proportional to Hz) another system for measuring frequency. The region of the infrared spectrum from 1200 to 700 cm -1 is called the fingerprint region. If we assume bonds are like springs, explain the frequencies in this table:.Order the bonds shown below in order of strength:.The lighter the weight, the higher (faster) the frequency of bouncing.The heavier the weight, the higher (faster) the frequency of bouncing.The weaker the spring, the higher (faster) the frequency of bouncing.The stronger the spring, the higher (faster) the frequency of bouncing.What happens to \(\nu\) if m is increased?.What happens to \(\nu\) if k is increased?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
